Hello everyone! My name is Taylor, I currently work as a certified pharmacy technician and through my experience have become very interested in the medical field. More specifically medications and how they effect the body. This is why I have chosen to study biomedical science here at ODU. This portfolio will show you my academic achievements, projects, and experiences. Thank you for visiting!
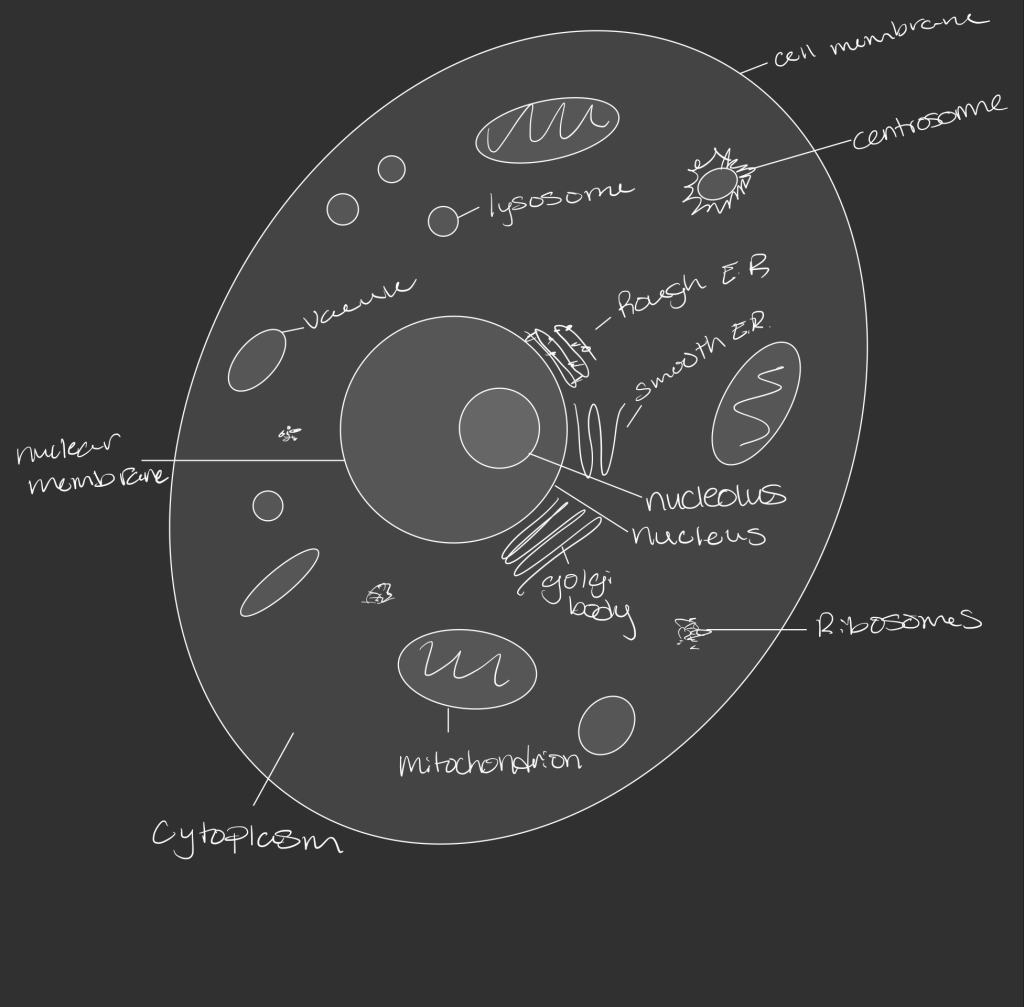
Here is my latest drawing of a cell!
Saturated Fat Biomolecule
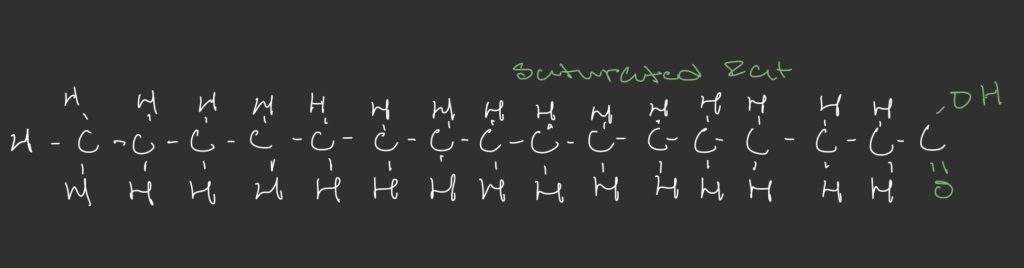

Scientific Literacy Essay
What is the incidence/rate of cardiac arrest in the US as a percentage of the population? What risk factors are associated with cardiac arrest? How often is it fatal?
The total percentage rate of cardiac arrest in the United States is 0.2% of the population. Breaking this down further, out-of-hospital cardiac arrest (OHCA) makes up about 0.11% of the population, while in-hospital cardiac arrest (IHCA) accounts for roughly 0.09%. These numbers show that cardiac arrest is a significant issue, whether it happens outside or inside a hospital setting.
There are many risk factors tied to cardiac arrest. A lot of them are related to cardiovascular diseases, such as coronary artery disease (CAD), heart failure, a prior heart attack (myocardial infarction), cardiomyopathy, and valvular heart disease. Beyond these, lifestyle and metabolic factors also play a big role. These include smoking, high blood pressure (hypertension), high cholesterol (hyperlipidemia), diabetes, obesity, and a sedentary lifestyle. In rarer cases, cardiac arrest can also be caused by things like drug overdoses or severe electrolyte imbalances, which disrupt the heart’s normal electrical activity.
The fatality rate of cardiac arrest is very high. For OHCA patients, the survival rate is only about 10%, meaning 90% of cases are fatal. For IHCA patients, the survival rate is slightly better at 25%, but that still means 75% of cases don’t survive. Without immediate medical intervention, cardiac arrest is almost always fatal. This highlights how critical it is to have quick access to CPR, defibrillators, and emergency medical services.
What is ischemia/reperfusion?
Ischemia happens when there’s a blockage or reduced blood flow to a tissue. This leads to a lack of oxygen and nutrients, which can cause injury or even cell death if it goes on too long. Reperfusion is when blood flow is restored to the tissue. While this is necessary to prevent further damage, it can also cause additional injuries. This is called reperfusion injury, and it happens because the sudden return of oxygen and nutrients can trigger inflammation and oxidative stress, which harm the tissue. This is a big concern in treatments for things like heart attacks or strokes, where restoring blood flow is essential but can also cause problems.
What is intercellular mitochondrial transfer? What does it suggest about mitochondrial transplant?
Intercellular mitochondrial transfer is when mitochondria move from one cell to another. This can happen through cell-to-cell connections, extracellular vesicles, or even cell fusion. Usually, this transfer happens in response to stress or injury, where damaged cells receive healthy mitochondria from nearby cells to help them recover. This has been seen in cells like neurons, cardiomyocytes (heart cells), and immune cells.
This process has some interesting implications for mitochondrial transplantation. It suggests that mitochondria are naturally able to move between cells and integrate into new ones, which could be useful for treating diseases. Here are some key points:
- Mitochondrial Therapy: Damaged cells could be repaired by giving them healthy mitochondria, which might help with conditions like ischemia or neurodegenerative diseases.
- Enhanced Cellular Recovery: Transplanted mitochondria could help restore ATP production, which is essential for cell function.
- Non-Invasive Delivery: In the future, mitochondria might be delivered through the bloodstream instead of being injected directly into tissues, making the process easier and less invasive.
- Challenges: There are still some hurdles to overcome, like making sure the transplanted mitochondria are compatible with the recipient cells and figuring out how long they’ll stay functional.
Overall, intercellular mitochondrial transfer shows that cells have a natural way of sharing and repairing their energy-producing machinery. This could lead to new treatments for a variety of diseases, but more research is needed to understand how it works and how to use it effectively.
Mitochondria are often called the “powerhouses” of our cells because they produce the energy needed for the body to function. This is especially important for cells like neurons and heart muscle cells, which use a lot of energy. When someone experiences cardiac arrest, blood flow to the brain and other organs stops, causing severe damage. In recent years, mitochondrial transplantation has been explored as a treatment to improve survival and brain recovery after cardiac arrest. This essay discusses how mitochondrial transplantation works, its effects on survival and recovery, how long the transplanted mitochondria stay in the body, and whether fresh or frozen mitochondria are more effective.
Mitochondrial transplantation is a process where healthy mitochondria are introduced into cells that have been damaged, such as after an ischemic event like cardiac arrest. These transplanted mitochondria can help restore energy production and reduce stress on the cells. Research shows that mitochondria can be successfully transplanted into neural cells, and they can continue to function properly. In studies, transplanted mitochondria have been shown to integrate into neurons, improving cellular function. This is especially important in the brain, where neurons rely heavily on mitochondria to work properly. When mitochondrial function is disrupted, as it is during cardiac arrest, it can cause cell death. By restoring mitochondrial function, transplantation can help protect neurons, improve brain function, and reduce brain injury.
Cardiac arrest stops blood flow to vital organs like the brain and heart, which leads to serious tissue damage. After someone is resuscitated, the goal is to limit further damage and help them recover. Mitochondrial transplantation has shown promise in improving recovery by boosting energy production in damaged cells. Studies indicate that mitochondrial transplantation can improve survival rates after cardiac arrest by helping cells produce the energy they need, which reduces cell death and supports healing.
In addition to improving survival, mitochondrial transplantation has positive effects on health after cardiac arrest. For example, it helps lower blood lactate levels, which rise after cardiac arrest due to anaerobic metabolism. When mitochondrial function is restored, the body can return to normal energy production, reducing lactate levels. Mitochondrial transplantation also helps reduce lung edema, a common complication after resuscitation. This happens because healthy mitochondria improve energy production in lung cells, which reduces inflammation and prevents fluid buildup in the lungs.
Mitochondrial transplantation can also improve glucose metabolism and enhance blood flow in the brain. By restoring mitochondrial function in cells that control blood flow, it helps improve circulation to the brain, ensuring that the brain gets the oxygen and nutrients it needs to heal.
One important question is how long transplanted mitochondria stay in the body and continue to work. Research shows that transplanted mitochondria can remain in the tissues for several days to weeks. However, their presence may decrease over time, especially in severely damaged tissues. Despite this, the immediate benefits of mitochondrial transplantation can still be significant, particularly in the critical period after resuscitation.
Fresh mitochondria are typically preferred for transplantation. Freezing mitochondria can cause damage, such as rupturing their membranes, which makes it harder for them to work properly. While freezing techniques have improved, fresh mitochondria still perform better when transplanted. However, future advancements in freezing methods may make frozen mitochondria a more viable option.
Mitochondrial transplantation shows great potential as a treatment to improve survival and brain recovery after cardiac arrest. By restoring mitochondrial function, it helps protect cells from damage, improves energy production, and supports recovery, especially in critical organs like the brain and heart. Although transplanted mitochondria may not last forever, their immediate benefits can be significant. Fresh mitochondria are currently the best option for transplantation, but future improvements in freezing techniques may change this. Overall, mitochondrial transplantation offers a hopeful approach for improving recovery after cardiac arrest and other injuries.
End-of-Term Reflection – Cell Biology and Its Relevance to Pharmacy
Throughout this cell biology course, some of the most meaningful information I learned was that insulin signaling pathway and how it regulates glucose uptake in cells. This topic linked to my pharmacy interests, especially since I had previously learned about how insulin and other antidiabetic medications manage diabetes. To understand how insulin binds to its receptor and activates a cascade of IRS proteins with the GLUT4 transporters allowed me to conceptualize how medications like metformin or insulin analogs function at the cellular level. In addition, I was able to understand the underlying mechanisms that result in insulin resistance and Type 2 diabetes. Overall, I have grown more confident to apply cellular mechanics to a real-world therapeutic problem because of the extra knowledge I gained. My experience in this course further demonstrated for me the importance of cell biology in understanding drug development and mechanisms and it ultimately has helped me grow and mature as a pharmacy student, particularly in an analytical perspective.