A monoclonal antibody that I found that is currently used in medicine today is Basiliximab. It is an immunosuppressive drug that prevents organ rejection for a patient who has received a kidney transplant. Rejection from a kidney transplant occurs when your body doesn’t accept the new kidney and fights against it (Cleveland Clinic, 2023). The immune system perceives the new kidney as a threat to one’s body. Although the donor kidney may be a perfect match, your body recognizes the kidney as a foreign object and tries to destroy it. Immunosuppressants are medications that aid in the treatment and prevention of a kidney transplant rejection. These anti-rejection drugs block one’s immune system from damaging the new kidney. There are also two forms of kidney transplant rejection: acute and chronic. Acute kidney transplant rejection can happen quickly within the first few weeks or year of the transplant. Chronic kidney transplant rejection occurs slowly and over several years. Displaying chronic rejection of a new kidney also typically leads to kidney damage due to the body’s immune system constantly fighting the new kidney. Some symptoms of kidney transplant rejection include high fever, flu-like symptoms, sudden weight gain, and less frequent urination. Basiliximab is among one of the different types of immunosuppressant drugs given to kidney transplant patients. Its antibody class is IgG, specifically IgG1k.
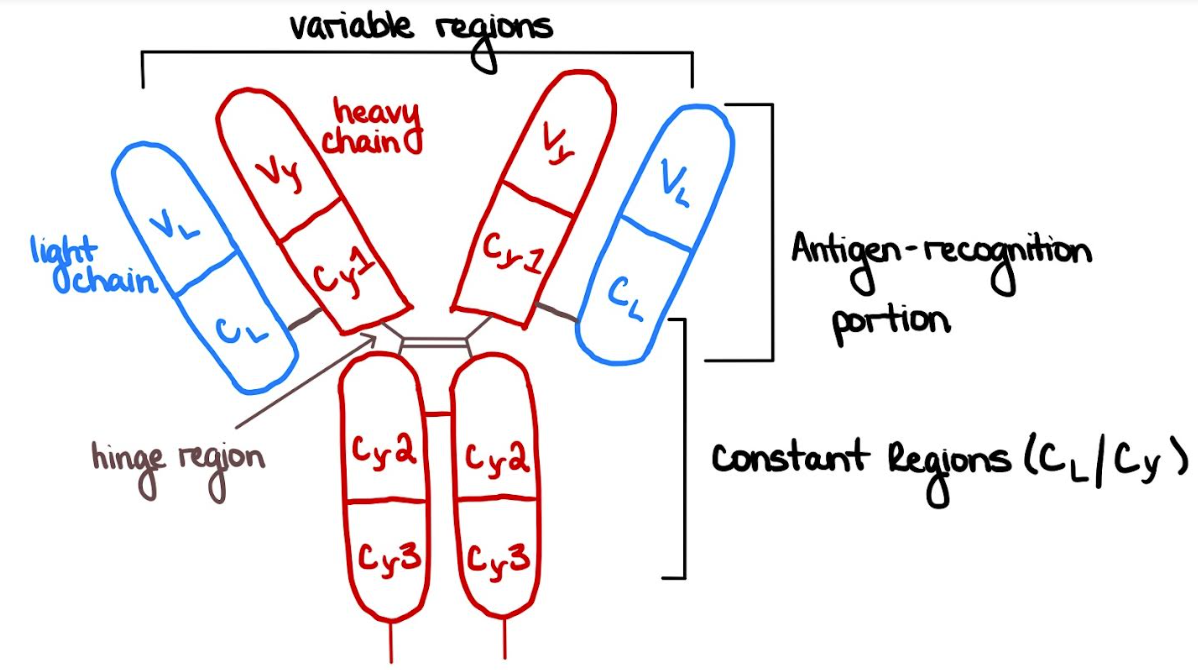
Figure 1: Brief Sketch of IgG Structure
Basiliximab is a glycoprotein that is a recombinant chimeric monoclonal antibody. A chimeric antibody occurs when one fuses the variable regions from one species with the constant regions of another species. The creation of the Basiliximab began from the fermentation of a myeloma cell line of a mouse. It was genetically engineered to express plasmids that contained a mouse’s light and heavy variable region genes and a human’s light and heavy constant region genes, thus classifying the biologic as a chimeric antibody. Geneticists also created this cell line to have the genes encode the RFT5 antibody that would bind selectively to the IL-2R (Interleukin-2 Receptor) α-subunit. The receptor is also known as the CD25 antigen. Due to its high affinity to the α-subunit of the IL-2 receptor, the drug prevents the formation of the IL-2R binding site on the surface of activated T-lymphocytes. Basiliximab also functions as a competitive antagonist of IL-2. Inhibiting the IL-2-mediated activation of lymphocytes allows Basiliximab to interrupt the cellular immune response involved with rejection, such as cell activation, cytokine release, and proliferation (Onrust & Wiseman, 1999). Since Basiliximab inhibits the critical pathway in T-cell activation, it prevents one’s body from attacking the foreign kidney. With its ability to hinder the immune response by the blockage of the IL-2R α-chain, Basiliximab also impedes the body from exhibiting any signs of rejection after renal transplantation.